Proud to represent Shantha College of Allied Health Sciences at the First National Conference organized by Acharya Institute of Allied Health Sciences.
A great opportunity to learn, interact with experts, and gain valuable insights in healthcare.




Shantha College of Pharmacy celebrated National Pharmacy Education Day – 2026 by organizing Pharma Anveshan 2026 on 6th March 2026 at the college campus. The program was conducted in commemoration of the birth anniversary of Mahadeva Lal Schroff, known as the Father of Pharmacy Education in India.
The event highlighted the theme “Future Pharma Ecosystem: Fostering Synergy among Academia, Industry, Research, Regulatory & Practice.” Students and faculty members actively participated, making the program a meaningful celebration of pharmacy education and its role in advancing healthcare.




Shantha College of Allied Health Sciences successfully conducted a seminar on “Cancer Awareness: From Knowledge to Prevention” on 28th February 2026 at the SGI Auditorium.
The session was delivered by Dr. Usha G Shenoy, Assistant Professor, Department of Physiology, Sri Devaraj Urs Academy of Higher Education & Research. The seminar focused on cancer prevention, early detection, risk factors, and the importance of awareness in reducing cancer incidence.
The program was organized under the guidance of the management and academic leadership and was well received by students and faculty members. The session concluded with an emphasis on promoting awareness and preventive healthcare practices in society.









Visit to: Group Pharma, Malur
Date: 18/02/2026
Class: B.Pharm 1st Semester
Number of Students: 50
Industry Representative: Mr. Vijay
Faculty Coordinator: Mr. Neela Madhav S
On 18th February 2026, 50 students of B.Pharm 1st Semester visited Group Pharma, Malur as part of our academic industrial exposure program. The main objective of the visit was to introduce first semester students to the basic functioning of a pharmaceutical manufacturing unit, particularly in the production of liquid orals and toothpaste formulations. The visit was coordinated by Mr. Neela Madhav S, and the industrial guidance was provided by Mr. Vijay.
The session began with a brief introduction about the company profile, its manufacturing facilities, and product range. Mr. Vijay explained the importance of Good Manufacturing Practices (GMP), hygiene, safety measures, and regulatory compliance in pharmaceutical industries.
During the plant tour, students were shown the manufacturing processes of:
Liquid Orals Manufacturing
Preparation of syrup base
Mixing and homogenization of ingredients
Filtration and in-process quality control
Bottle filling, sealing, labeling, and packaging
The importance of maintaining correct measurements, pH, viscosity, and microbial safety was explained in a simple and understandable manner for first semester students.
Toothpaste Manufacturing
Mixing of abrasives, binders, active ingredients, and flavoring agents
Homogenization to achieve smooth consistency
Vacuum processing to remove air bubbles
Tube filling, crimping, and final packaging
Students also visited the Quality Control (QC) Laboratory, where basic tests such as pH testing, viscosity measurement, and quality checks were demonstrated. The Quality Assurance (QA) department explained the importance of documentation and maintaining batch records.
Mr. Vijay emphasized workplace safety, clean room standards, and teamwork in pharmaceutical manufacturing. An interactive session was conducted at the end, where students clarified their basic doubts about industrial processes and career opportunities.
Overall, the industrial visit to Group Pharma, Malur was highly informative and beneficial for all 50 students of B. Pharm 1st Semester. We sincerely thank the management of Group Pharma, especially Mr. Vijay, and our faculty coordinator Mr. Neela Madhav S for organizing and guiding us throughout this valuable learning experience.


Visit to: Phytosol
Date: 13/02/2026
Class: B.Pharm 5th Semester
Number of Students: 50
Industry Representative: Ms. Sindhu (Phytosol)
Faculty Coordinator: Mr. Neela Madhav S
On 13th February 2026, 50 students of B.Pharm 5th Semester visited Phytosol as part of our academic curriculum to gain practical exposure to pharmaceutical industry operations. The visit was coordinated by Mr. Neela Madhav S, and all the industrial instructions and explanations were provided by Ms. Sindhu from Phytosol.
The visit commenced with a brief introduction about the company profile, its product range, and its commitment towards quality and regulatory compliance. Ms. Sindhu explained the importance of Good Manufacturing Practices (GMP), safety measures, and adherence to Standard Operating Procedures (SOPs) in pharmaceutical manufacturing.
During the plant tour, students were taken through various departments including:
Production Department: The manufacturing process, equipment handling, workflow system, and hygiene maintenance were clearly demonstrated.
Quality Control (QC) Laboratory: Students were introduced to analytical instruments and testing procedures used to ensure product quality and compliance.
Quality Assurance (QA) Department: Documentation practices such as batch manufacturing records, validation processes, and quality monitoring systems were explained.
Packaging Unit: The process of labeling, inspection, and final packaging of finished products was shown and described in detail.
Ms. Sindhu emphasized the importance of clean room standards, proper storage of raw materials, safety protocols, and documentation in maintaining product quality. The session concluded with an interactive discussion where students clarified their doubts regarding career opportunities and industrial expectations.
Overall, the industrial visit to Phytosol was highly informative and beneficial for all 50 students. We sincerely thank the management of Phytosol and especially Ms. Sindhu for guiding us throughout the visit, as well as Mr. Neela Madhav S for coordinating and organizing this valuable learning experience








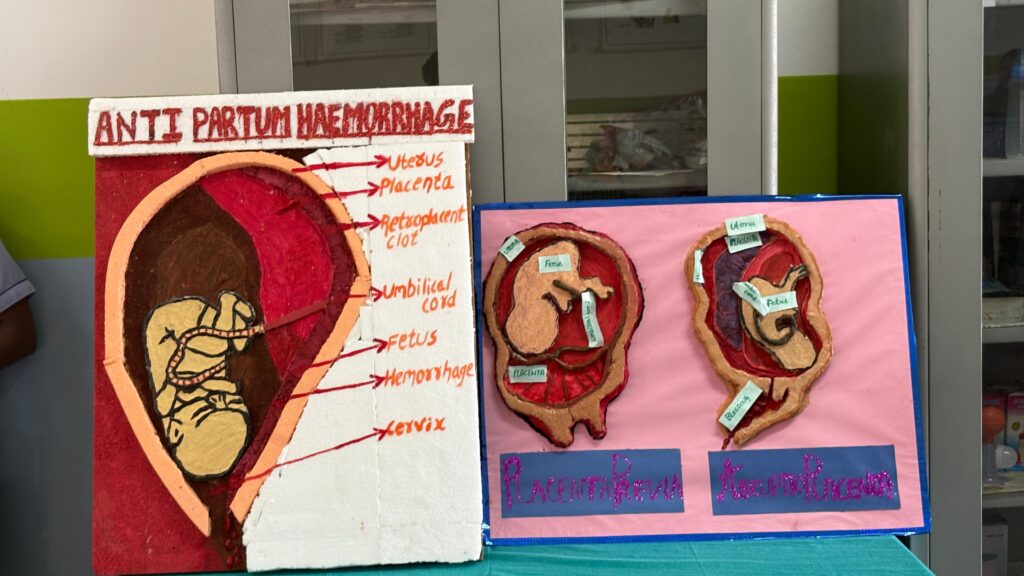
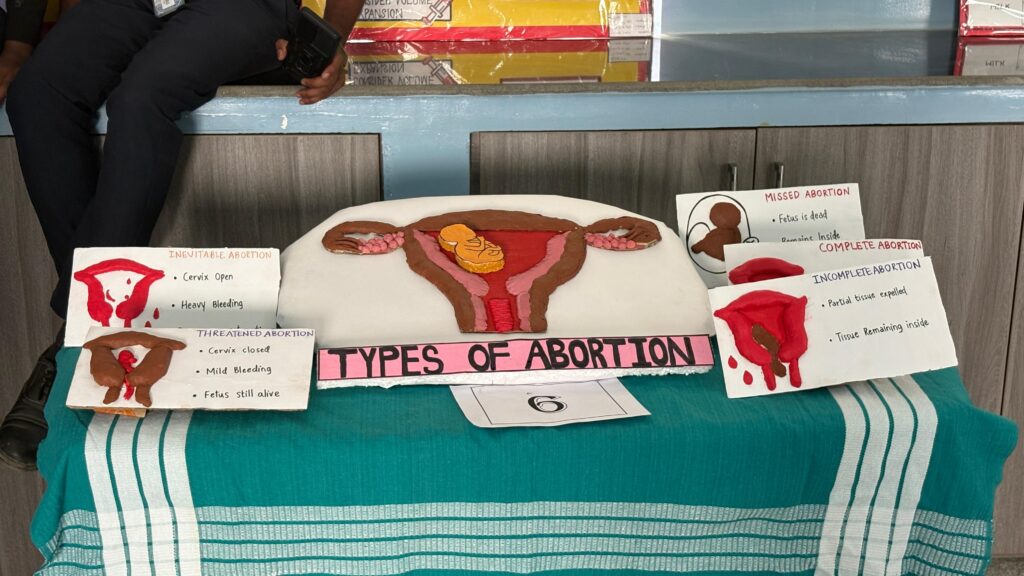
The Department of Obstetrics and Gynecology (OBG) Nursing successfully organized an OBG Model Expo on the college campus with the objective of enhancing students’ practical knowledge and encouraging innovative learning. The expo provided a platform for students to demonstrate their understanding of maternal and reproductive health through creative and informative models.
Students actively participated by presenting models on various important topics such as Fertilization, physiology of first stage of labour, mechanism of labour, abortion and its type, fetal circulation, assisted vaginal delivery , Leopold maneuvers and antepartum haemorrhage. The models were designed using innovative materials and clearly labeled to enhance conceptual understanding.
Each group explained their model confidently, highlighting its clinical relevance and practical application in maternal health care. Judges evaluated the models based on concept clarity, creativity, presentation, and practical utility.
The expo not only strengthened theoretical knowledge but also improved communication skills, teamwork, and confidence among students. It created awareness about safe motherhood practices and the importance of quality obstetric and gynecological care.
The OBG Model Expo was a great success and proved to be an effective teaching–learning strategy, making complex concepts easier to understand through visual and practical demonstration.